the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Djaffa Mountains guereza (Colobus guereza gallarum) abundance in forests of the Ahmar Mountains, Ethiopia
Afework Bekele
Anagaw Atickem
Dietmar Zinner
The size and density of a population are essential parameters in primate ecology and conservation. Such information, however, is still scarce for many forest primate species. The Djaffa Mountains guereza (Colobus guereza gallarum) is an endemic Ethiopian taxon for which data about its distribution and population size are missing. Therefore, we aimed to estimate the abundance and population size of the Djaffa Mountains guereza in four forests in the Ahmar Mountains southeast of the Ethiopian Rift Valley. We conducted line-transect surveys in the forests. Within an area of 183 km2, we sampled 19 transects covering a distance of 75.9 km. We encountered 73 guereza clusters which most likely represent social groups. Since the detection distances and cluster sizes did not differ among the four forests, we applied a conventional distance sampling (CDS) model and estimated a population density of 20.6 clusters per square kilometer, i.e., 109.6 individuals per square kilometer or 20 061 individuals within the complete study area. This abundance is relatively high compared to other C. guereza taxa. However, given that the habitat and population of C. g. gallarum are already highly fragmented, further monitoring of the population and exploration of the possibilities of reconnecting its habitat should be priorities for the conservation of this taxon.
- Article
(2879 KB) - Full-text XML
-
Supplement
(671 KB) - BibTeX
- EndNote
Large parts of the global biodiversity are threatened by extinction, including many primate species. Most primate species (93 % of the species) are experiencing population declines (Estrada and Garber, 2022), and the International Union for Conservation of Nature (IUCN) lists over 65 % of primate species as “Vulnerable”, “Endangered”, or “Critically Endangered” (Fernández et al., 2022). The reasons for this negative trend are generally well-known and include the destruction, fragmentation, and conversion of primate habitats; hunting; and illegal trade (Estrada et al., 2017). Future human population growth, agricultural expansion, and climate change are expected to accelerate the decline of primate populations (Estrada et al., 2020; Estrada and Garber, 2022; Pinto et al., 2023). To develop species-specific conservation strategies and/or to monitor implemented conservation measures, estimating densities and abundances of populations is essential (Jachmann, 2001; Marques, 2001; Keeping and Pelletier, 2014; Kiffner et al., 2022a). This is particularly important for threatened species living in already human-modified landscapes (Cavada et al., 2016). Animal population surveys are therefore an essential contribution to the successful conservation of species (Ogutu et al., 2006; Santini et al., 2022).
In recent decades, Ethiopia has experienced a severe loss of forest habitats in almost all regions of the country and thus the primary habitats for forest-dependent species, including several primate taxa (Yalden et al., 1977). One primate taxon that is most likely at extinction risk is the Djaffa Mountains guereza (Colobus guereza gallarum Neumann, 1902, hereinafter referred to as DMG; Fig. 1) because, in its supposed range, only a few forested areas remain (Kufa et al., 2022). DMG is a black-and-white colobus endemic to the Arsi and Ahmar Mountains in Ethiopia (Groves, 2001; Fashing and Oates, 2013, 2019; Zinner et al., 2019; Butynski and De Jong, 2022). However, its exact distribution is uncertain. In particular, information on the range boundaries between DMG and C.g. guereza is missing. The occurrence of DMGs west of the Arsi and Ahmar Mountains, e.g., in Munessa Forest, Wondo Genet Forest, Dale Forest, and Bale Mountains National Park, is questionable (Petros et al., 2018a, b, c; Menbere and Mekonen, 2019; Mekonen and Hailemariam, 2021). At the very least, these western populations do not carry DMG-specific mitochondrial haplotypes (Zinner et al., 2019; Tesfaye et al., 2021). Given the dramatic loss and degradation of forests in its range as well as its likely limited distribution, the DMG is most likely facing a severe risk of extinction (Kufa et al., 2022). However, due to poor knowledge of its population size and distribution, the DMG is listed as “Data Deficient” by the IUCN (Fashing and Oates, 2019).

Figure 1Male Djaffa Mountains guereza (Colobus guereza gallarum) in Hades Forest, 2021 (photo: Chala A. Kufa).
In this study, we estimated the population density and size of DMGs in four of the remaining forests in the Ahmar Mountains of Ethiopia. We used line-transect distance sampling, which has been widely used to estimate the densities of diurnal arboreal primates in tropical forests (González-Solís et al., 2001; Buckland et al., 2010; Höing et al., 2013; Leca et al., 2013; Araldi et al., 2014; Omifolaji et al., 2020; Kiffner et al., 2022a, b). This method is relatively simple, rapid, and cost-effective (Buckland et al., 2001; Thomas et al., 2010). Furthermore, it is also used for sparsely distributed populations for which sampling needs to be efficient, populations that occur in well-defined clusters and at low or medium cluster density, and populations that are detected through a flushing response (Buckland et al., 2001).
2.1 Ethical statement
This research adhered to the legal requirements of Ethiopia, and there was no animal handling. Permission to carry out the study was obtained from the Department of Zoological Sciences, Addis Ababa University, and the Oromia Forest and Wildlife Enterprise.
2.2 Study sites
We conducted our study in the Ahmar Mountains, a mountain range of the Ethiopian Highlands south of the Rift Valley in the Oromia regional state of Ethiopia (Fig. 2). The topography of the Ahmar Mountains is characterized by plateaus, rugged dissected mountains, deep valleys, gorges, and plains (Abdala et al., 2017). The climate of the study area receives a bimodal rainfall distribution (Fig. S1a–d), with a short rainy season between February and May, long rains between July and September, and a long dry period between October and January.
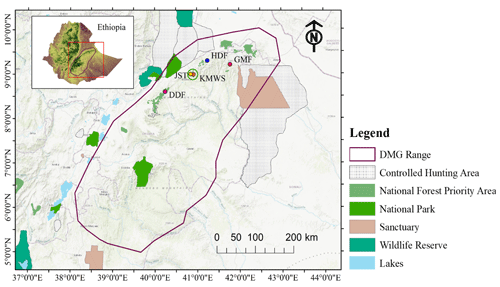
Figure 2Geographic position of the study area in the Ahmar Mountains, Ethiopia, and locations of the four forest sites where we did the line-transect surveys on Djaffa Mountains guereza (DMG): Dindin Forest (DDF), Jallo Kuni-Muktar Wildlife Sanctuary (JKF, comprising two sites nearby: Jallo Sorroro Torgam – JST – and Kuni-Muktar Wildlife Sanctuary – KMWS), Hades Forest (HDF), and Gara Muleta Forest (GMF) (source of protected area shapefiles: UNEP-WCMC and IUCN, 2023).
Originally, dry evergreen Afromontane forest dominated the region (Bishaw, 2001; Friis et al., 2010; Asefa et al., 2020). However, most parts of the Ahmar Mountains are now covered by wooded grasslands, with stands of exotic tree species such as Eucalyptus and bushland (Friis et al., 2010). The remaining forests in the region are often fragmented and degraded due to unsustainable use and persistent drought (Abdala et al., 2017).
Table 1Characteristics of each study site in the Ahmar Mountains, Ethiopia: geographic coordinates in decimal degrees; elevation (m a.s.l.); size (km2); mean (±SD) annual precipitation (mm); mean (±SD) annual temperature (∘C).
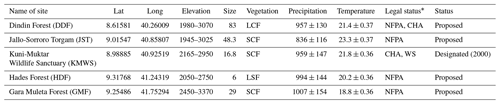
NFPA: national forest priority area; CHA: controlled hunting area; WS: wildlife sanctuary; LSF: large forest fragment (1–10 km2); SCF: small continuous forest (11–50 km2); LCF: large continuous forest (> 50 km2). ∗ Source: UNEP-WCMC and IUCN (2023).
For our survey, we selected four forests (Table 1; Fig. 3) where DMGs had been reported in previous studies (Zinner et al., 2019; Kufa et al., 2022) and where we confirmed their presence in a pilot study (unpublished data). These forests have been managed at the regional level by the Oromia Forest and Wildlife Enterprise (OFWE), which is advocating the preservation and protection of the natural forest through a participatory forest management approach. As a restoration practice, Juniperus procera, Cupressus lusitanica, Hagenia abyssinica, and Croton macrostachyus have been planted by the OFWE in collaboration with the local community.
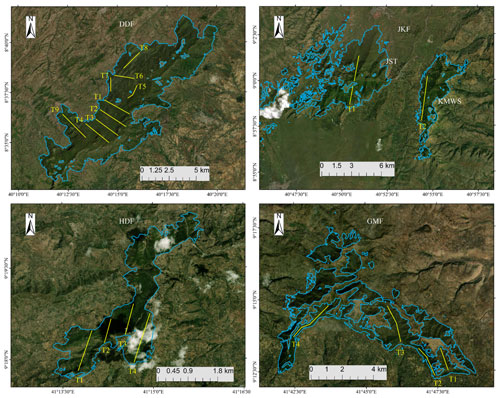
Figure 3Positions of transects (yellow lines) within the four forests used for DMG surveys from 2020 to 2021: DDF, JKF, HDF, and GMF. The maps show the extents of the study sites for each forested area. Base map: ©Google Earth.
The four forests differ slightly in their ecologies (Table 1). DDF is one of the remnants of the natural forests, marking a large continuous forest in the eastern part of the country. The dominant woody vegetation found in the forest includes Juniperus procera, Olinia rochetiana, Maytenus addat, Maytenus undata, Podocarpus falcatus, Myrsine africana, Olea europaea, Maesa lanceolata, Myrsine melanophloeos, and Schefflera volkensii (Shibru and Balcha, 2004).
JKF is a concession-controlled hunting area that comprises two fragmented forests, JST and KMWS. The concession area is represented by woody tree species such as M. lanceolata, J. procera, C. macrostachyus, Rhus glutinosa, and Acacia abyssinicus (Reshad et al., 2020).
HDF consists mainly of dry Afromontane forest comprising 40–48 woody species (Teketay, 1997; Atomsa and Dibbisa, 2019). The dominant tree species are C. macrostachyus, H. abyssinica, Schefflera abyssinica, and Prunus africana.
GMF harbors diverse flora, with about 361 species of vascular plants, of which 45 (13 %) are endemic to Ethiopia (Teketay, 1996). The southern side of Gara Muleta is covered by a multi-story mixed deciduous forest dominated by Ekebergia capensis and P. africana associated with intermediate and lower-story species that include Bersama abyssinica, C. macrostachyus, Dioscorea schimperiana, Erythrina brucei, H. abyssinica, Nuxia congesta, O. africana, and S. abyssinica (Teketay, 1996).
2.3 Survey method, design, and data collection
As a method of estimating abundance, we conducted line-transect surveys in four forest sites in the range of DMG in the Ahmar Mountains, employing a conventional distance sampling (CDS) approach (Buckland et al., 2001). This approach only considers the perpendicular distance to estimate the detection probability, thus in turn inferring the population size and estimating the density of a detected animal (Buckland et al., 1993, 2001).
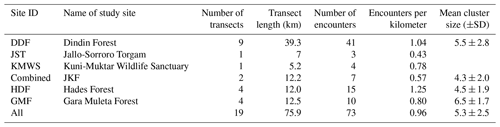
We generated 19 transect lines (Fig. 3; range: 1.8–7.0 km) on topological maps of the four forest fragments using ArcGIS 10.4.1 in a random sampling design (Thomas et al., 2010; Buckland et al., 2015). Of the 19 transects, 9 were in DDF, 2 in Jallo-Kuni Muktar Wildlife Sanctuary, 4 in Hades Forest, and 4 in Gara Muleta Forest, covering a total distance of 75.9 km (Table 2). We positioned the transects in parallel wherever possible, with 1 km between adjacent transects, except for transects 3 and 4 of GMF, where the distance is approximately 5 km.
Table 2Number of transects per study site, lengths of transects, encounter rates, and mean cluster sizes per study site.
We did the fieldwork from December 2020 to September 2021. From 07:00 to 18:00 Eastern African Time, a team of three well-trained persons led by CAK walked slowly along each transect (1 km h−1) (Peres, 1999; Plumptre et al., 2013). We assumed that our target species had daily travel distances similar to a closely related taxon, C. g. occidentalis (62 to 1360 m; Fashing, 2001a). We surveyed each transect once per season.
Colobus monkeys are relatively easy to detect because their movements, loud calls, and pelage make them conspicuous (Fig. 1). They often indicate their presence with loud calls that may be heard for more than 1 km. For each encounter, we recorded the (1) transect ID, (2) date and time of the sighting, (3) weather conditions, (4) sighting location along the transect using the Global Positioning System (Garmin® GPSmap 76CSx), (5) number of individuals (cluster size), (6) perpendicular distance (visually estimated), and (7) initial detection cue (auditory or visual). The personnel estimated the perpendicular distance after they were carefully trained in how to estimate distances and until the errors in distance estimation were reduced to less than 2 m. We visually estimated the perpendicular distances to the center of each cluster at their first detection (Kiffner et al., 2022a).
The home range size of C. guereza ranges from 0.075 to 1 km2 (Fashing, 2011), and daily travel distances vary from 390 to 600 m (Fashing, 2011). Core areas are usually defended, and groups keep a certain distance from each other (von Hippel, 1996). We therefore defined individuals > 50 m apart as members of different clusters (Teelen, 2007; Kiffner et al., 2022b).
2.4 Data analyses
We conducted our survey in four forests and two seasons, and since forest ecology and season (more or less good visibility) can affect detection distance (perpendicular distance), we first tested whether forest ID or season (dry and wet) affected the perpendicular distances. Second, we compared cluster sizes among the four forests and tested whether cluster size affected the perpendicular distances since larger groups might be easier to detect (shorter perpendicular distances). We did these tests in R 3.4.1 (R Core Team, 2022) and found no effects of forest ID, season, or cluster size on detection distances (see the Appendix; Fig. S2a–e). We therefore combined all our encounters in a single analysis.
Since we recorded the perpendicular distances to the nearest meter, we subsequently ordered them into four distance classes to better fit the detection function: 0–10 m, > 10–20 m, > 20–30 m, and > 30–50 m. We had only two encounters with perpendicular distances larger than 50 m (one with 75 m and a cluster size of 5 and one with 100 m and a cluster size of 2). We therefore truncated the distribution at 50 m to increase the robustness of the detection function (Buckland et al., 1993, 2001). To establish the detection models on a large enough sample size since 60–80 detections are advised for consistently producing accurate density estimations (Peres, 1999; Buckland et al., 2001) and our sample sizes do not support separate fitting of the detection function to the data in each stratum of the forest, we combined all data across the study sites for fitting a detection function.
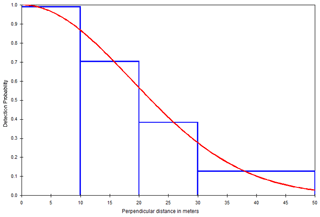
Using the pooled encounters across the forests, we fitted four candidate detection models to check the performance of these functions (uniform, half-normal, hazard rate, and negative exponential) in the conventional distance sampling (CDS) in Distance 7.3 Release 2 (Thomas et al., 2010) (Table 3; Fig. S3a–d). This determines the basic model shape. We considered three series expansions or adjustment terms, i.e., cosine, Hermite polynomial, and simple polynomial (Table 3), to make the models more robust.
Table 3Results from fitting different detection models for DMG across the four forest sites during the survey periods. These models are arranged by differences in Akaike's information criterion (ΔAIC*) between each candidate model and the model with the lowest AIC value. The key functions are uniform (UN), half-normal (HN), hazard rate (HR), and negative exponential (NE). The adjustment terms are cosine (CS), simple polynomial (SP), and Hermite polynomial (HP). The number of parameters (“np”) is shown for each model. “Pa” is the estimated detection probability; “ESW” is the estimated strip width in meters, “D” is the population density, and “CV” is the coefficient of variation. Numbers in parentheses denote the 95 % confidence intervals.
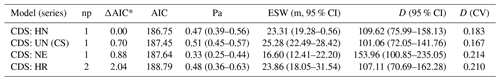
We selected the best model using the Akaike information criterion (AIC) (Akaike, 1973) and retained the half-normal key function with no adjustment term (ΔAIC = 0.0; AIC = 186.75; Table 3). In addition, we considered the chi-squared goodness-of-fit test and the visual fit of the models as additional model selection criteria (Buckland et al., 2001). We used the chi-squared test to compare the number of observations in a given distance interval to the number expected under the fitted detection function (Table S1). Then, we computed the probability of detecting a cluster given it is in the covered area, which is used to correct the density estimation (i.e., the number of groups per square kilometer) across the study periods and to determine the effective strip width (ESW, which is the distance at which as many objects are seen) (Thomas et al., 2010).
We computed the mean size of clusters in the population, the density of clusters, the population density, and the abundance of clusters for all the data combined. Since predicted cluster size estimation based on the size-biased regression method indicated warnings, we used the mean of the observed clusters as the basis for expected cluster size estimation. We also computed relative abundance (i.e., an index of abundance, usually presented as an encounter rate (ER) of objects recorded per unit of distance; Campbell et al., 2016; Fewster et al., 2009). We finally estimated the population size of the study taxon by multiplying the global lumped population density by the total area of the studied forests (183 km2). We quantified parameter estimates of uncertainty or variance using the standard error (SE), percent of the coefficient of variation (%CV), and 95 % confidence intervals (CIs) in the analytic variance estimation method in the distance.
3.1 Encounter rate and cluster size
In total, we encountered DMGs 73 times (Table 2), with the highest encounter rate in HDF (1.25 per kilometer), followed by DDF (1.04 per kilometer), GMF (0.80 per kilometer), and JKF (0.57 per kilometer). The overall encounter rate was 0.96 per kilometer. The number of DMG individuals per cluster varied from 1 to 15 (N=73), with an overall mean cluster size of 5.3±2.5 (mean±SD).
3.2 Modeling the detection function
The result of the model selection suggested a model with a half-normal key function and no simple polynomial adjustment as the best detection model (Table 3). The chi-squared goodness-of-fit tests show that a detection function model provides an adequate fit to the grouped distance data (χ2=0.1682, df = 2, P=0.9193; Table S1). Figure 4 depicts the fitted detection function averaged over the observed perpendicular distance for the half-normal model. Histograms of detected distances show higher detections close to the line transect, fitting the assumption of distance sampling analyses (Fig. 4).
3.3 Population size and density estimate of Djaffa Mountains guerezas
We estimated the group density as 20.6 clusters per square kilometer (95 % CI: 14.5–29.3; %CV = 17.5; df = 35.1) and the population density of DMGs as 109.6 individuals per square kilometer (95 % CI: 76.0–158.1; %CV = 18.3 %; df = 42.3) (Table 4). The estimated total population size for the complete study area of 183 km2 was 20 061 individuals (95 % CI: 13 907–28 938; %CV = 18.3).
Table 4Parameter estimate analysis and inference for DMGs across the forest fragments during the survey periods using the best model selected, i.e., half-normal key . “A(1)” is the ith parameter in the estimated probability density function (pdf); “f(0)” is , the effective detection area for line transects, which is the value of the pdf at zero for line transects; “p” is the estimated detection probability; “ESW” is the effective strip width for line transects W*p; “n/L” is the encounter rates; “DS” is the estimate of the density of clusters; “E(S)” is the estimate of the expected value of a cluster size; “D” is the estimate of the density of animals; and “N” is the estimate of the number of animals in a specified area.
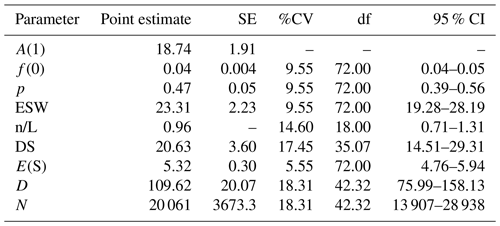
During our field surveys, we encountered 73 clusters of DMGs. The estimated population parameters of DMGs were 20.6 clusters per square kilometer, 109.6 individuals per square kilometer, or 20 061 individuals for the complete study area of 183 km2. The mean cluster size of DMGs (5.3 individuals) did not differ among the four study sites. If one assumes that the cluster size corresponds to the size of their social groups, this figure is at the lower end of reported average group sizes for other C. guereza subspecies (5.4 to 19; Fashing, 2011). It remains an open question whether the relatively low group size of DMG is taxon-specific or is caused by any ecological factor.
Our encounter rate (0.96 clusters per kilometer) is comparable to other colobus sites (1.2 clusters per kilometer, Kakamega Forest, Kenya, Fashing and Cords, 2000; 1.65 clusters per kilometer, central Ethiopia, Yazezew et al., 2022). Similarly, our population density estimate (109.6 individuals per square kilometer) fits into the range of population densities found at other sites (4.9 to 150 individuals per square kilometer, with one outlier of 315 individuals per square kilometer; Table 5), although one has to be careful when directly comparing population densities from different sites and with different ecologies, and most importantly when different census methods have been applied (Spaan et al., 2019; Kiffner et al., 2022b). Since there is a lack of baseline data, we are also not able to establish with certainty whether the population of DMG has declined, increased, or remained unaffected due to changes that occurred in the fragmented forests of the Ahmar Mountains.
Table 5Population parameters of the African black-and-white colobus.
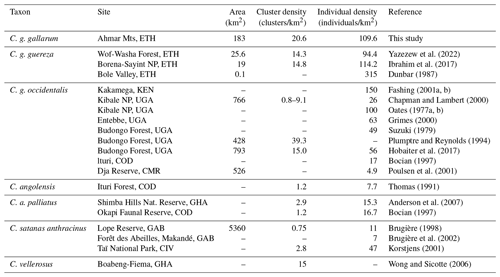
CMR: Cameroon; CIV: Côte d'Ivoire; COD: Democratic Republic of Congo; ETH: Ethiopia; GAB: Gabon; GHA: Ghana; KEN: Kenya; TZA: Tanzania; UGA: Uganda.
Generally, guerezas are ecologically relatively flexible, and they can survive even in small forest fragments (e.g., just a few trees surrounding a church in Ethiopia) (Dunbar and Dunbar, 1974; Fashing et al., 2019). They can also subsist in parks or can be found in tiny forest remnants in towns and are, in general, tolerant of the presence of humans (Yalden et al., 1977). Given the density of DMGs within the four study forests, conservation concern seems not to be a low population density per se but might be more the small range and the low number of suitable forests within the range of DMGs (Kufa et al., 2022). Habitat suitability models revealed that only 1336 km2 (1.8 %) of the 75 307 km2 study area consist of a highly suitable habitat for DMGs under current climate conditions (Kufa et al., 2022). Given that the species occurs mainly at higher altitudes, climate change can have additional negative effects on the habitat of DMGs, especially if the vegetation belts are “pushed” uphill, similar to what is expected for other high-altitude species in Ethiopia (Ahmed et al., 2023).
Because of their assumed limited geographic distribution (Zinner et al., 2019; Kufa et al., 2022) and therefore an assumed relatively small population size, DMG is most likely facing a higher risk of extinction than C. g. guereza. Also, the remnant forests where DMGs are found are isolated, making genetic exchange among the local populations of DMG difficult if not impossible. This can lead to an increase in inbreeding and a loss of genetic diversity. We therefore suggest a population genetic study to assess the genetic status of DMG. A comprehensive survey to collect samples for genetic analysis (noninvasive sampling, e.g., fecal material) could be the next step. We further suggest determining the geographic distribution of C. g. gallarum, because several forests that constitute a suitable habitat for DMG are unexplored, e.g., in the Arba-Gugu Mountains. It would also be important to collect data along the common distribution border between DMGs and other guereza taxa and to check for possible sympatry and hybridization. Finally, we recommend that conservation management programs focus on reconnecting forest fragments to re-establish dispersal routes among currently isolated local populations of DMGs. However, this should be accompanied by a public awareness campaign and discussions with the stakeholders involved.
All raw data can be provided by the corresponding author upon reasonable request.
The supplement related to this article is available online at: https://doi.org/10.5194/pb-10-13-2023-supplement.
CAK and AA conceived and designed the field surveys. CAK performed the fieldwork. CAK and DZ analyzed the data. CAK, AA, AB, and DZ wrote the manuscript.
At least one of the (co-)authors is a member of the editorial board of Primate Biology. The peer-review process was guided by an independent editor, and the authors also have no other competing interests to declare.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. While Copernicus Publications makes every effort to include appropriate place names, the final responsibility lies with the authors.
We thank the Oromia Forest and Wildlife Enterprise (OFWE) of the Hararghe district for providing permission to conduct the field surveys in the designated forests. We thank the residents and OFWE officials, especially Jebessa Fituma and Olana Badada, who assisted in data collection in the four forests. We also thank the two anonymous reviewers and a topical editor for their valuable and constructive comments.
CAK has been supported by the Jana Robeyst Trust Fund, Wondelgem, Belgium (RPR Gent: BE 0656.646.943/2019CE13), and the Thematic Research Fund of the Department of Zoological Sciences, Addis Ababa University, Ethiopia (AAU Thematic project 11, 2020).
This paper was edited by Stefan Schlatt and reviewed by two anonymous referees.
Abdala, T., Eshetu, G., and Worku, A.: A participatory assessment of forest biodiversity resources and level of threat in Hararge Area, eastern Ethiopia, Int. J. Sci. Tech. Soc., 5, 67–73, https://doi.org/10.11648/j.ijsts.20170504.13, 2017.
Ahmed, S. A., Chala, D., Kufa, C. A., Atickem, A., Bekele, A., Svenning, J. C., and Zinner, D.: Distribution and extent of suitable habitat for geladas (Theropithecus gelada) in the Anthropocene. bioRxiv, 2023.2008.2010.552774, https://doi.org/10.1101/2023.08.10.552774, 2023.
Akaike, H.: Information theory as an extension of the maximum likelihood principle, in Proceedings of the Second International Symposium on Information Theory, edited by: Petrov, B. N. and Csaki, F., Akademiai Kiado, Budapest, Hungaria, 267–281, 1973.
Anderson, J., Rowcliffe, J. M., and Cowlishaw, G.: The Angola black-and-white colobus (Colobus angolensis palliatus) in Kenya: Historical range contraction and current conservation status, Am. J. Primatol., 69, 664–680, https://doi.org/10.1002/ajp.20377, 2007.
Araldi, A., Barelli, C., Hodges, K., and Rovero, F.: Density estimation of the endangered Udzungwa red colobus (Procolobus gordonorum) and other arboreal primates in the Udzungwa Mountains using systematic distance sampling, Int. J. Primatol., 35, 941–956, https://doi.org/10.1007/s10764-014-9772-6 , 2014.
Asefa, M., Cao, M., He, Y., Mekonnen, E., Song, X., and Yang, J.: Ethiopian vegetation types, climate and topography, Plant Diversity, 42, 302–311, https://doi.org/10.1016/j.pld.2020.04.004, 2020.
Atomsa, D. and Dibbisa, D.: Floristic composition and vegetation structure of Ades forest, Oromia regional state, West Hararghe zone, Ethiopia, Trop. Plant Res., 6, 139–147, https://doi.org/10.22271/tpr.2019.v6.i1.020, 2019.
Bishaw, B.: Deforestation and land degradation in the Ethiopian Highlands: A strategy for physical recovery, Northeast Afr. Studies, 8, 7–25, https://doi.org/10.1353/nas.2005.0014, 2001.
Bocian, C.: Niche Separation of Black-and-White Colobus Monkeys (Colobus angolensis and C. guereza) in the Ituri Forest, PhD thesis, City University of New York, New York, USA, 220 pp., 1997.
Brugière, D.: Population size of the black colobus monkey Colobus satanas and the impact of logging in the Lope Reserve, central Gabon, Biol. Conserv., 86, 15–20, https://doi.org/10.1016/S0006-3207(98)00015-9, 1998.
Brugière, D., Gautier, J. P., and Gautier-Hion, A.: Primate diet and biomass in relation to vegetation compostion and fruiting phenology in rain forest in Gabon, Int. J. Primatol., 23, 999–1024, https://doi.org/10.1023/A:1019693814988, 2002.
Buckland, S. T., Anderson, D. R., Burnham, K. P., and Laake, J. L.: Distance Sampling: Estimating Abundance of Biological Populations, Chapman and Hall, London, UK, 446 pp., ISBN: 0412426609, 1993.
Buckland, S. T., Anderson, D. R., Burnham, K. P., Laake, J. L., Borchers, D. L., and Thomas, L.: Introduction to Distance Sampling. Estimating Abundance of Biological Populations, Oxford University Press, Oxford, UK, 448 pp., ISBN: 9780198509271, 2001.
Buckland, S. T., Plumptre, A. J., Thomas, L., and Rexstad, E. A.: Design and analysis of line transect surveys for primates. Int. J. Primatol., 31, 833–847, https://doi.org/10.1007/s10764-010-9431-5, 2010.
Buckland, S. T., Rexstad, E. A., Marques, T. A., and Oedekoven, C. S.: Distance Sampling: Methods and Applications, Springer, Cham, Switzerland, 277 pp., https://doi.org/10.1007/978-3-319-19219-2, 2015.
Butynski, T. M. and De Jong, Y. A.: Conservation of Africa's colobine monkeys (Cercopithecidae, Colobinae) with taxonomic and biogeographic considerations, in: The Colobines: Natural History, Behaviour and Ecological Diversity, edited by: Matsuda, I., Grueter, C. C., and Teichroeb, J. A., Cambridge University Press, Cambridge, UK, 342–393, https://doi.org/10.1017/9781108347150.022, 2022.
Campbell, G., Head, J., Junker, J., and Nekaris, K. A. I.: Primate abundance and distribution: background concepts and methods, in: An Introduction to Primate Conservation, edited by: Wich, S. A., and Marshall, A. J., Oxford University Press, Oxford, UK, 79–110, https://doi.org/10.1093/acprof:oso/9780198703389.003.0006, 2016.
Cavada, N., Barelli, C., Ciolli, M., and Rovero, F.: Primates in human-modified and fragmented landscapes: The conservation relevance of modelling habitat and disturbance factors in density estimation, PLoS ONE, 11, e0148289, https://doi.org/10.1371/journal.pone.0148289, 2016.
Chapman, C. A. and Lambert, J. E.: Habitat alteration and the conservation of African primates: Case study of Kibale National Park, Uganda, Am. J. Primatol., 50, 169–185, https://doi.org/10.1002/(SICI)1098-2345(200003)50:3<169::AID-AJP1>3.0.CO;2-P, 2000.
Dunbar, R. I. M.: Habitat quality, population dynamics, and group composition in colobus monkeys (Colobus guereza), Int. J. Primatol., 8, 299–329, https://doi.org/10.1007/BF02737386, 1987.
Dunbar, R. I. M. and Dunbar, E. P.: Ecology and population dynamics of Colobus guereza in Ethiopia, Folia Primatol., 21, 188–208, https://doi.org/10.1159/000155600, 1974.
Estrada, A. and Garber, P. A.: Principal drivers and conservation solutions to the impending primate extinction crisis: Introduction to the Special Issue, Int. J. Primatol., 43, 1–14, https://doi.org/10.1007/s10764-022-00283-1, 2022.
Estrada, A., Garber, P. A., Rylands, A. B., Roos, C., Fernandez-Duque, E., Di Fiore, A., Nekaris, K. A. I., Nijman, V., Heymann, E. W., Lambert, J. E., Rovero, F., Barelli, C., Setchell, J. M., Gillespie, T. R., Mittermeier, R. A., Arregoitia, L. V., de Guinea, M., Gouveia, S., Dobrovolski, R., Shanee, S., Shanee, N., Boyle, S. A., Fuentes, A., MacKinnon, K. C., Amato, K. R., Meyer, A. L. S., Wich, S., Sussman, R. W., Pan, R., Kone, I., and Li, B.: Impending extinction crisis of the world's primates: Why primates matter, Sci. Adv., 3, e1600946, https://doi.org/10.1126/sciadv.1600946, 2017.
Estrada, A., Garber, P. A., and Chaudhary, A.: Current and future trends in socio-economic, demographic and governance factors affecting global primate conservation, PeerJ, 8, e9816, https://doi.org/10.7717/peerj.9816, 2020.
Fashing, P. J.: Activity and ranging patterns of guerezas in the Kakamega Forest: Intergroup variation and implications for intragroup feeding competition, Int. J. Primatol., 22, 549–577, https://doi.org/10.1023/A:1010785517852, 2001a.
Fashing, P. J.: Male and female strategies during intergroup encounters in guerezas (Colobus guereza): evidence for resource defense mediated through males and a comparison with other primates, Behav. Ecol. Sociobiol. 50, 219–230, https://doi.org/10.1007/s002650100358, 2001b.
Fashing, P. J.: African Colobine monkeys: Their behaviour, ecology, and conservation, in: Primates in Perspective, edited by: Campbell, C. J., Fuentes, A., MacKinnon, K. C., Bearder, S. K., and Stumpf, R. M., New York: Oxford University Press, 203–229, ISBN: 978-0-19-539043-8, 2011.
Fashing, P. J. and Cords, M.: Diurnal primate densities and biomass in the Kakamega Forest: An evaluation of census methods and a comparison with other forests, Am. J. Primatol., 152, 139–152, https://doi.org/10.1002/(SICI)1098-2345(200002)50:2<139::AID-AJP4>3.0.CO;2-N, 2000.
Fashing, P. J. and Oates, J. F.: Colobus guereza, in: Mammals of Africa: vol 2 Primates, edited by: Butynski, T. M., Kingdon, J., and Kalina, J., Bloomsbury, London, UK, 111–119, ISBN: 978-1-4081-2252-5, 2013.
Fashing, P. J. and Oates, J. F.: Colobus guereza ssp. gallarum. The IUCN Red List of Threatened Species 2019: e.T5150A17983175, https://doi.org/10.2305/IUCN.UK.2019-3.RLTS.T5150A17983175.en, 2019.
Fashing, P. J., Tesfaye, D., Yazezew, D., and Oates, J. F.: Colobus guereza ssp. guereza (errata version published in 2020). The IUCN Red List of Threatened Species 2019: e.T136896A164179773, https://doi.org/10.2305/IUCN.UK.2019-3.RLTS.T136896A164179773.en, 2019.
Fernández, D., Kerhoas, D., Dempsey, A., Billany, J., McCabe, G., and Argirova, E.: The current status of the world's primates: Mapping threats to understand priorities for primate conservation, Int. J. Primatol., 43, 15–39, https://doi.org/10.1007/s10764-021-00242-2, 2022.
Fewster, R. M., Buckland, S. T., Burnham, K. P., Borchers, D. L., Jupp, P. E., Laake, J. L., and Thomas, L.: Estimating the encounter rate variance in distance sampling, Biometrics, 65, 225–236, 2009.
Friis, L., Demissew, S., and van Breugel, P.: Atlas of the Potential Vegetation of Ethiopia, Biologiske Skrifter, 58, 1–307, 2010.
González-Solís, J., Guix, J. C., Mateos, E., and Llorens, L.: Population density of primates in a large fragment of the Brazilian Atlantic rainforest, Biodivers. Conserv. 10, 1267–1282, https://doi.org/10.1023/A:1016678126099, 2001.
Grimes, K. H.: Guereza Dietary and Behavioural Patterns at the Entebbe Botanical Gardens, PhD thesis, Calgary University, Calgary, Canada, 146 pp., http://hdl.handle.net/1880/40711 (last access: 13 October 2023), 2000.
Groves, C. P.: Primate Taxonomy, Washington, DC, Smithsonian Institution Press, 350 pp., ISBN: 1-56098-872-X, 2001.
Hobaiter, C., Samuni, L., Mullins, C., Akankwasa, W. J., and Zuberbühler, K.: Variation in hunting behaviour in neighbouring chimpanzee communities in the Budongo forest, Uganda, PLoS ONE, 12, 1–17, https://doi.org/10.1371/journal.pone.0178065, 2017.
Höing, A., Quinten, M. C., Indrawati, Y. M., Cheyne, S. M., and Waltert, M.: Line transect and triangulation surveys provide reliable estimates of the density of Kloss' gibbons (Hylobates klossii) on Siberut Island, Indonesia, Int. J. Primatol., 34, 148–156, https://doi.org/10.1007/s10764-012-9655-7, 2013.
Ibrahim, H., Bekele, A., and Yazezew, D.: Population structure and feeding ecology of guereza (Colobus guereza) in Borena-Sayint National Park, northern Ethiopia, Int. J. Biodiver. Conserv., 9, 323–333, https://doi.org/10.5897/IJBC2017.1114, 2017.
Jachmann, H.: Estimating Abundance of African Wildlife: An Aid to Adaptive Management, J. Wildl. Manag., 1391, 40–47, https://doi.org/10.1007/978-1-4615-1381-0, 2001.
Keeping, D. and Pelletier, R.: Animal density and track counts: understanding the nature of observations based on animal movements, PLoS ONE, 9, e96598, https://doi.org/10.1371/journal.pone.0096598, 2014.
Kiffner, C., Kioko, J., Butynski, T. M., de Jong, Y. A., and Zinner, D.: Population dynamics of the Manyara monkey (Cercopithecus mitis manyaraensis) and vervet monkey (Chlorocebus pygerythrus) in Lake Manyara National Park, Tanzania, Primate Biol., 9, 33–43, https://doi.org/10.5194/pb-9-33-2022, 2022a.
Kiffner, C., Paciência, F. M. D., Henrich, G., Kaitila, R., Chuma, I. S., Mbaryo, P., Knauf, S., Kioko, J., and Zinner, D.: Road-based line distance surveys overestimate densities of olive baboons, PLoS ONE, 17, 1–18, https://doi.org/10.1371/journal.pone.0263314, 2022b.
Korstjens, A. H.: The Mob, the Secret Sorority, and the Phantoms: An Analysis of the Socio-Ecological Strategies of the Three Colobines of Taï, PhD thesis, Utrecht University, 173 pp., ISBN: 90-393-2752-1, 2001.
Kufa, C. A., Bekele, A., and Atickem, A.: Impacts of climate change on predicted habitat suitability and distribution of Djaffa Mountains Guereza (Colobus guereza gallarum, Neumann 1902) using MaxEnt algorithm in Eastern Ethiopian Highland, Glob. Ecol. Conserv., 35, e02094, https://doi.org/10.1016/j.gecco.2022.e02094, 2022.
Leca, J. B., Gunst, N., Rompis, A., Soma, G., Putra, I. A., and Wandia, I. N.: Population density and abundance of ebony leaf monkeys (Trachypithecus auratus) in West Bali National Park, Indonesia, Primate Conserv., 26, 133–144, https://doi.org/10.1896/052.026.0106, 2013.
Marques, F. F. C.: Estimating Wildlife Distribution and Abundance from Line Transect Surveys Conducted from Platforms of Opportunity, PhD thesis, University of St Andrews, 157 pp., http://hdl.handle.net/10023/3727 (last access: 13 October 2023), 2001.
Mekonen, S. and Hailemariam, M.: Population size, group composition and feeding ecology of the endemic and endangered Colobus guereza gallarum in Harenna Forest, Harenna Buluk District, south east Ethiopia, Egypt. Acad. J. Biol. Sci., 13, 65–75, https://doi.org/10.21608/eajbsz.2021.193040, 2021.
Menbere, I. P. and Mekonen, S.: Why Colobus monkeys (Colobus guereza) are attracted to human settlements, and urinate when they come across humans in their territory and in predator dominant places? Observation from Bale Mountains National Park, in Ethiopia, Adv. Life Sci. Tech., 75, 9–13, 2019.
Oates, J. F.: The guereza and its food, in: Primate ecology: Studies of feeding and ranging behaviour in lemurs, monkeys and apes, edited by Clutton-Brock, T. H., Academic Press, London, 275–321, ISBN: 0-12-176850-3, 1977a.
Oates, J. F.: The social life of a black-and-white colobus monkey, Colobus guereza, Z. Tierpsychol, 45, 1–60, 1977b.
Ogutu, J. O., Bhola, N., Piepho, H. P., and Reid, R.: Efficiency of strip-and line-transect surveys of African savanna mammals, J. Zool., 269, 149-160, https://doi.org/10.1111/j.1469-7998.2006.00055.x, 2006.
Omifolaji, J. K., Ikyaagba, E. T., Alarape, A. A., Ojo, V. A., Modu, M., Lateef, L. F., Adeyemi, M. A., Ahmad, S., and Xiaofeng, L.: Estimates of Demidoff's galago (Galagoides demidovii) density and abundance in a changing landscape in the Oban Hills, Nigeria, Hystrix, the Italian J. Mammal., 31, 117–122, https://doi.org/10.4404/hystrix-00342-2020, 2020.
Peres, C. A.: General guidelines for standardizing line-transect surveys of tropical forest primates, Neotrop. Primates, 7, 11–16, 1999.
Petros, I., Mekonen, S., Gena, H., and Mesfin, Y.: Diurnal activity patterns and social behaviour of Colobus guereza gallarum in Bale Mountains National Park, Southeast Ethiopia, Species, 19, 15–22, 2018a.
Petros, I., Mekonen, S., Gena, H., and Mesfin, Y.: Feeding and ranging ecology of Colobus guereza gallarum in Bale Mountains National Park, southeast Ethiopia, J. Biodivers. Endangered Species, 6, 007, https://doi.org/10.4172/2332-2543.S2-007, 2018b.
Petros, I., Mekonen, S., Gena, H., and Mesfin, Y.: Population status, distribution, and threats of Colobus guereza gallarum in Bale Mountains National Park, southeastern Ethiopia, Int. J. Nat. R. Ecol. Manag., 3, 39–45, https://doi.org/10.11648/j.ijnrem.20180303.12, 2018c.
Pinto, M. P., Beltrão-Mendes, R., Talebi, M., and de Lima, A. A.: Primates facing climate crisis in a tropical forest hotspot will lose climatic suitable geographical range, Sci. Rep., 13, 641, https://doi.org/10.1038/s41598-022-26756-0, 2023.
Plumptre, A. J. and Reynolds, V.: The effect of selective logging on the primate populations in the Budongo Forest Reserve, Uganda, J. Appl. Ecol., 31, 631–641, https://doi.org/10.2307/2404154, 1994.
Plumptre, A. J., Sterling, E. J., and Buckland, S. T.: Primate census and survey techniques, in: Primate Ecology and Conservation: A Handbook of Techniques, edited by: Sterling, E. J., Bynum, N., and Blair, M. E., Oxford University Press, Oxford, UK, 10–26, https://doi.org/10.1093/acprof, 2013.
Poulsen, J. R., Clark, C. J., and Smith, T. B.: Seed dispersal by a diurnal primate community in the Dja Reserve, Cameroon, J. Trop. Ecol., 17, 787–808, 2001.
R Core Team: R: A language and environment for statistical computing, R Foundation for Statistical Computing, Vienna, Austria, https://www.r-project.org/ (last access: 13 October 2023), 2022.
Reshad, M., Mohammed, M., and Beyene, A.: Distribution and importance value index of woody species under different successional stages at Jello-Muktar dry Afromontane forest, South-eastern Ethiopia, Am. J. Agr. Forest. 8, 1–8, https://doi.org/10.11648/j.ajaf.20200801.11, 2020.
Santini, L., Benítez-López, A., Dormann, C. F., and Huijbregts, M. A.: Population density estimates for terrestrial mammal species, Glob. Ecol. Biogeog., 31, 978–994, https://doi.org/10.1111/geb.13476, 2022.
Shibru, S. and Balcha, G.: Composition, structure and regeneration status of woody species in Dindin Natural Forest, Southeast Ethiopia: An implication for conservation, Eth. J. Biol. Sci., 3, 15–35, 2004.
Spaan, D., Ramos-Fernández, G., Schaffner, C. M., Smith-Aguilar, S. E., Pinacho-Guendulain, B., and Aureli, F.: Standardizing methods to estimate population density: an example based on habituated and unhabituated spider monkeys, Biodivers. Conserv., 28, 847–862, https://doi.org/10.1007/s10531-018-01696-2, 2019.
Suzuki, A.: The variation and adaptation of social groups of chimpanzees and black and white colobus monkeys, in: Primate Ecology and Human Origins, edited by: Bernstein, I. S. and Smith, E. O., Garland STPM Press, New York, 153–174, ISBN: 978-0824070809, 1979.
Teelen, S.: Primate abundance along five transect lines at Ngogo, Kibale National Park, Uganda, Am. J. Primatol., 69, 1030–1044, https://doi.org/10.1002/ajp.20417, 2007.
Teketay, D.: Floristic composition of Gara Muleta and Kundudo Mountains, south-eastern Ethiopia: implications for the conservation of biodiversity, in: The Biodiversity of African Plants: Proceedings XIVth AETFAT Congress 22–27 August 1994, Wageningen, The Netherlands, Springer Netherlands, 345–350, ISBN: 978-94-010-6613-6, 1996.
Teketay, D.: Seedling populations and regeneration of woody species in dry Afromontane forests of Ethiopia, Forest Ecol. Manag., 98, 149–165, https://doi.org/10.1016/S0378-1127(97)00078-9, 1997.
Tesfaye, D., Fashing, P. J., Atickem, A., Bekele, A., and Stenseth, N. C.: Feeding ecology of the Omo River guereza (Colobus guereza guereza) in habitats with varying levels of fragmentation and disturbance in the southern Ethiopian Highlands, Int. J. Primatol., 42, 64–88, https://doi.org/10.1007/s10764-020-00189-w, 2021.
Thomas, L., Buckland, S. T., Rexstad, E. A., Laake, J. L., Strindberg, S., Hedley, S. L., Bishop, J. R. B., Marques, T. A., and Burnham, K. P.: Distance software: Design and analysis of distance sampling surveys for estimating population size, J. Appl. Ecol., 47, 5–14, https://doi.org/10.1111/j.1365-2664.2009.01737.x, 2010.
Thomas, S. C.: Population densities and patterns of habitat use among anthropoid primates of the Ituri Forest, Zaire, Biotropica, 23, 68–83, https://doi.org/10.2307/2388690, 1991.
UNEP-WCMC and IUCN: Protected Planet: The World Database on Protected Areas (WDPA) and World Database on Other Effective Area-based Conservation Measures (WD-OECM) [Online], Cambridge, UK: UNEP-WCMC and IUCN, https://www.protectedplanet.net/en (last access: 13 October 2023), 2023.
von Hippel, F. A.: Interactions between overlapping multimale groups of black and white colobus monkeys (Colobus guereza) in the Kakamega Forest, Kenya, Am. J. Primatol., 38, 193–209, https://doi.org/10.1002/(SICI)1098-2345(1996)38:3<193::AID-AJP1>3.0.CO;2-U, 1996.
Wong, S. N. P. and Sicotte, P.: Population size and density of Colobus vellerosus at the Boabeng-Fiema Monkey Sanctuary and surrounding forest fragments in Ghana, Am. J. Primatol., 68, 465–476, https://doi.org/10.1002/ajp.20242, 2006.
Yalden, D., Largen, M., and Kock, D.: Catalogue of the mammals of Ethiopia, 3. Primates, Monitore Zoologico Italiano (Ital. J. Zool.) NS Suppl. 9, 1–52, https://doi.org/10.1080/03749444.1977.10736841, 1977.
Yazezew, D., Bekele, A., Fashing, P. J., Nguyen, N., Moges, A., Ibrahim, H., Burke, R. J., Eppley, T. M., and Mekonnen, A.: Population size and habitat preference of the Omo River guereza (Colobus guereza guereza) in a multi-habitat matrix in the central highlands of Ethiopia, Primates, 63, 151–160, https://doi.org/10.1007/s10329-022-00972-8, 2022.
Zinner, D., Tesfaye, D., Stenseth, N. C., Bekele, A., Mekonnen, A., Doeschner, S., Atickem, A., and Roos, C.: Is Colobus guereza gallarum a valid endemic Ethiopian taxon?, Primate Biol., 6, 7–16, https://doi.org/10.5194/pb-6-7-2019, 2019.